Active Surveillance
What is surveillance?
Surveillance is an essential public health tool. It is the systematic collection, analysis, and dissemination of data on diseases/conditions of public health importance so that appropriate action can be taken.
Public Health surveillance takes data into action. Surveillance data can help determine the burden of disease, assess interventions, and be used to improve clinical practice and health policy. Surveillance data usually generates hypotheses and actions while research tests hypotheses.
What is active surveillance?
Surveillance can be either passive or active:
Passive surveillance involves issuing the case definition to an existing health care network and then waiting for case reports to come in. It is inexpensive but limited in completeness and timeliness.
Active surveillance directly contacts health care providers or laboratories to request if they have seen cases. While this method is more labour intensive, it provides a more complete estimate of disease frequency and the opportunity for case-level review.
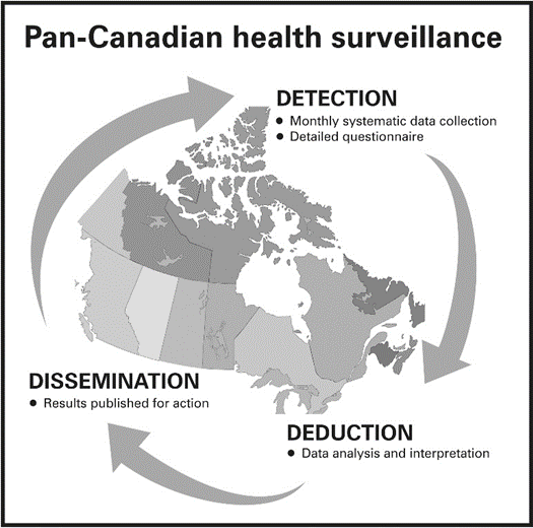
The Canadian Paediatric Surveillance Program (CPSP) is an active surveillance network of about 2700 paediatricians, paediatric subspecialists, and other medical professionals across Canada. The CPSP’s active surveillance model involves:
Detection: The CPSP uses two data collection mechanisms: multi-year studies and one-time surveys. For multi-year studies, CPSP participants complete monthly case notification forms. Participants who have seen a case are asked to complete a clinical questionnaire.
Deduction: CPSP study and survey investigators analyze the surveillance data and interpret it into new clinical and public health information.
Dissemination: CPSP study and survey results are published and communicated to those who need to know so that appropriate action can be taken.
How does the CPSP work?
Call for proposals
The CPSP encourages teams of investigators from across Canada to submit proposals for new surveillance studies or one-time surveys that address a rare paediatric health condition or an emerging public health issue.
Scientific review and selection of proposals
The CPSP’s Scientific Steering Committee assesses study and survey proposals against established evaluation criteria and each proposal undergoes a rigorous scientific review. The Committee selects the proposals that are of highest medical and public health importance, on topics for which there is limited information, and for which surveillance is the most appropriate means of collecting the data.
 Detection
Detection
For each approved study, the CPSP initiates data collection fromapproximately 2700 participating paediatricians and paediatric subspecialists across Canada by sending them the case definition and the study protocol. The study is also added to the monthly case notification form. This approach provides a uniform basis for reporting and increases awareness and knowledge of unusual or rare conditions.
Studies use a two-tiered data collection mechanism:
- Participants receive a case notification form each month asking if they have seen any cases from a list of conditions under surveillance. Participants notify the Program of the number of new cases seen in the last month or indicate if they have nothing to report. Participants are encouraged to report all cases, including suspect or probable cases. This sometimes leads to duplicate reporting but avoids missed cases. A reply of “no cases seen” is also valuable, as it sets the population denominator and provides more accurate data.
- Participants who report having seen a case are sent a clinical questionnaire to collect further information.* Once the completed clinical questionnaire is returned to the CPSP, all notifications of potential cases are assessed against the case definition. Duplicates or cases that don't meet the case definition are excluded.
*Note: CPSP participants in Quebec who report a case will only receive a clinical questionnaire to complete if they are reporting a case from an institution with research ethics board approval for the study in question.
Reminders are sent to participants who have not replied, which greatly improves response rates and the ascertainment of cases. The CPSP’s national average monthly case notification rate is around 80% and the response rate for the completion of clinical questionnaires is between 83 to 93%. These participation rates show the dedication of Canadian paediatricians and paediatric subspecialists to participating in the CPSP.
For one-time surveys, a survey questionnaire is developed by the survey team and approved by the Scientific Steering Committee. All CPSP participants are asked to complete the survey and response rates vary from between 25 and 50%.
Deduction
Both study and survey teams analyze the surveillance data and interpret it into new information that can be used by clinicians, researchers, and policy makers.
Dissemination
The CPSP shares information from its studies and one-time surveys with key stakeholders so that improvements can be made to medical care and health policies for children and youth. The CPSP shares its results through a variety of mechanisms, including the following:
- Publications in scientific, peer-reviewed journals
- Annual CPSP Results
- Highlights published in Paediatrics & Child Health
- Surveillance program articles
- ·Articles published in CPS Blog
- Poster and oral presentations
Privacy
The CPSP assures privacy and the confidentiality of all information provided to the Program. No direct patient identifiers are collected. Only anonymized data from patient charts is used and the CPSP has no direct contact with patients or their families. Should a CPSP study or survey result in publication, only aggregate data are reported and case counts of fewer than five are suppressed.
Limitations of surveillance
Surveillance serves an important purpose and provides rich clinical data that allows for a better understanding of the rare childhood diseases/conditions under study. As with any voluntary reporting surveillance system, the CPSP recognizes that its surveillance has some limitations:
- Reporting on minimum incidence rates can under-represent events in the population. For example, some cases may not be included in the surveillance totals because they presented to health care practitioners other than paediatricians, while others may live in rural or remote areas and are less likely to receive timely specialist care.
- Some data elements (e.g., laboratory investigations, pre-existing medical conditions) may not be available at the time of reporting and therefore may be absent from the surveillance totals.Every effort is made to ensure complete data capture and to handle missing data appropriately in the data analysis.
- Data from Quebec are incomplete. Due to Quebec legislation, detailed case-level data can only be included in the data analysis when reported from a centre with project-specific research ethics board approval.

